Smith & Nephew Hip Recall
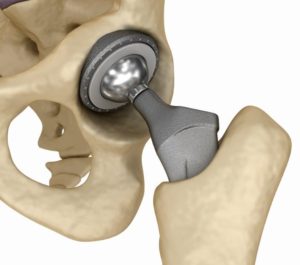 Smith & Nephew is a medical device manufacturer in London that provides their products worldwide used in hip, knee, and shoulder replacements. Their latest recall involving their hip replacement systems and the company voluntarily mailed out safety notice letters to inform the consumer on November 15, 2016.
Smith & Nephew is a medical device manufacturer in London that provides their products worldwide used in hip, knee, and shoulder replacements. Their latest recall involving their hip replacement systems and the company voluntarily mailed out safety notice letters to inform the consumer on November 15, 2016.
The devices involved in the recall are the modular neck hip prostheses of the Modular SMF and Modular REDAPT revision femoral hip systems. The joint replacement manufacturing giant made the decision to issue the hip system recalls because of, in their words, “a higher than anticipated complaint and adverse event trend.” This is not the first time the company has seen such complaints and adverse events in their history, but this latest one is due to their medical devices posing the danger to patients of metal debris being released by the implant into the blood. This can cause chromium and cobalt ion releases into the blood that can cause blood poisoning and also DNA damage (genotoxicity). Muscles and bones can be destroyed with these issues and the toxicity can carry over to other organs as well. The cobalt ions have caused heart muscle disease (cardiomyopathy). Metal poisoning occurs when there is loosening of the medical device and the bone and muscle surrounding the replacement deteriorate. Read more


 If you are like most, you are probably ready to be told any way to avoid sickness, as in listeria illness, over the upcoming holiday season.
If you are like most, you are probably ready to be told any way to avoid sickness, as in listeria illness, over the upcoming holiday season. Samsung seems to be having a bad 2016.
Samsung seems to be having a bad 2016.